Organ Models & Tissue Engineering
Multi-Organoid Patterning and Fusion (MOrPF)
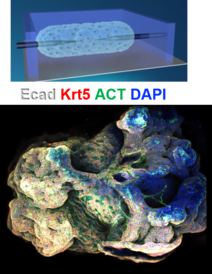
We have developed an approach, called Multi-Organoid Patterning and Fusion (MOrPF), for ‘up-sizing’ organoids: miniature collections of cells which mimic the behaviour of various organs and are promising tools for the study of human biology and disease. Our approach was used to culture and grow a ‘mini-airway’, the first time that a tube-shaped organoid has been developed without the need for any external support. Using a mould made of a specialised polymer, we were able to guide the size and shape of the mini-airway, grown from adult mouse stem cells, and then remove it from the mould when it reached the point where it could support itself. Whereas the organoids currently used in medical research are at the microscopic scale, the method developed by the Cambridge team could make it possible to grow life-sized versions of organs.
see video
https://www.youtube.com/watch?v=Ngfaq3j-vnk
https://doi.org/10.1002/advs.202003332
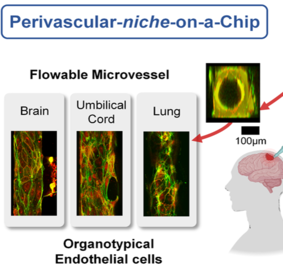
Microvessels on a chip
This research area focuses on developing models to recapitulate the microvasculature, and other key microenvironment factors for living cells in a fluidic device. Specifically, we apply our materials approach to study the motility of cancer cells when they interact with a vessel formed by the endothelial cells, to mimic the conditions of cancer invasion. We have also created a glomerulus-on-a-chip, for industry-relevant drug testing of kidney disease.
https://doi.org/10.1038/s41598-018-30776-0
DOI: 10.1039/D1LC00271F
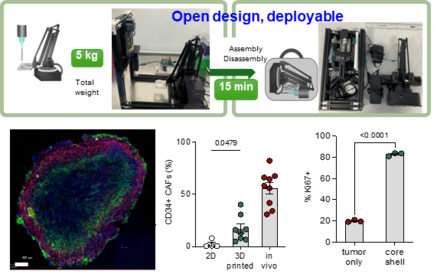
Deployable bioprinting of compartmental tumoroids with cancer associated fibroblasts for immune cell interactions
Realizing the translational impacts of three-dimensional (3D) bioprinting for cancer research necessitates innovation in bioprinting workflows which integrate affordability, user-friendliness, and biological relevance. Herein, we demonstrate 'BioArm', a simple, yet highly effective extrusion bioprinting platform, which can be folded into a carry-on pack, and rapidly deployed between bio-facilities. BioArm enabled the reconstruction of compartmental tumoroids with cancer-associated fibroblasts (CAFs), forming the shell of each tumoroid. The 3D printed core–shell tumoroids showed de novo synthesized extracellular matrices, and enhanced cellular proliferation compared to the tumour alone 3D printed spheroid culture. Further, the in vivo phenotypes of CAFs normally lost after conventional 2D co-culture re-emerged in the bioprinted model. Embedding the 3D printed tumoroids in an immune cell-laden collagen matrix permitted tracking of the interaction between immune cells and tumoroids, and subsequent simulated immunotherapy treatments. Our deployable extrusion bioprinting workflow could significantly widen the accessibility of 3D bioprinting for replicating multi-compartmental architectures of tumour microenvironment, and for developing strategies in cancer drug testing in the future.
Link to publication: DOI 10.1088/1758-5090/acb1db
Link to outreach video: https://youtu.be/H6LiCXH6dVs